GLP-1 drugs have become one of the fastest-growing product categories in healthcare. That commercial success has also been followed by a sharp rise in litigation. What started as scattered product liability filings has developed into a fast-growing federal mass tort focused first on alleged gastrointestinal injuries and now on alleged vision-loss claims. ¹
Why GLP-1s Are Becoming a Legal Issue
Plaintiffs allege that manufacturers failed to provide adequate warning of serious adverse effects associated with GLP-1 drugs, particularly gastrointestinal injuries such as gastroparesis, ileus, intestinal obstruction, and pseudo-obstruction. A newer group of cases alleges that certain GLP-1 drugs contributed to non-arteritic anterior ischemic optic neuropathy, or NAION, a condition associated with sudden vision loss.²
At this stage, GLP-1 litigation is not yet defined by verdicts or major settlement benchmarks. It is being defined instead by rapid case growth, a large and expanding user base, and injury allegations serious enough to keep plaintiff firms interested. For now, GLP-1s look like an emerging tort risk, but not yet a mature one.¹
A Fast-Growing Litigation Landscape
According to the U.S. Judicial Panel on Multidistrict Litigation, federal GLP-1 MDL cases increased by more than 120% between March 2025 and March 2026. ³
Pending federal GLP-1 MDL actions, March 2024–March 2026 ³
Takeaway: The federal GLP-1 docket moved from a small emerging filing group in 2024 to a much larger federal litigation in 2026, and it now includes a separate vision-loss MDL in addition to the gastrointestinal docket. ³ That kind of growth suggests the litigation has moved beyond isolated claims. Once a drug-related docket reaches the thousands, the question is no longer just whether individual plaintiffs can find counsel. It becomes a question of whether the litigation can support coordinated expert development, plaintiff advertising, and a long enough runway to become a more durable liability issue. ³
The Scale Behind the Risk
The size of the underlying market helps explain why the litigation has drawn so much attention. KFF reported in November 2025 that 18% of U.S. adults said they had ever taken a GLP-1 drug and 12% said they were currently taking one.⁴ Eli Lilly’s CEO David Ricks has publicly suggested that more than 20% of Americans could eventually use GLP-1s.
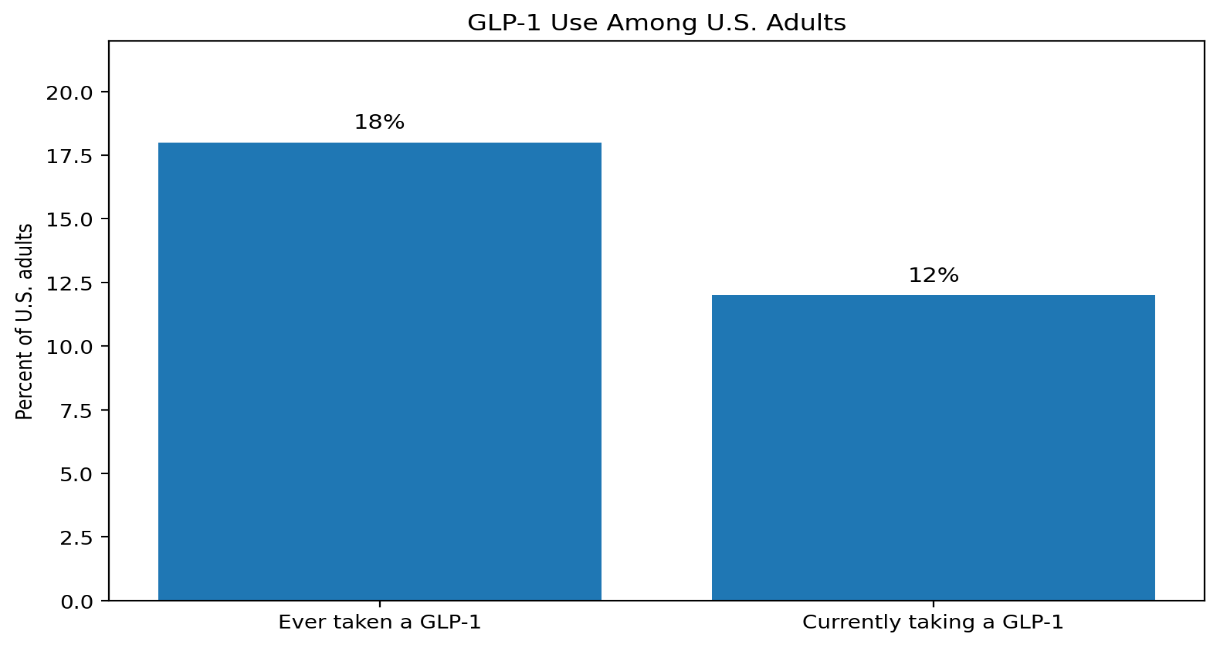
That user base may grow further as oral forms become available. Reuters reported in January 2026 that Novo Nordisk expected oral pills to capture more than one-third of the global GLP-1 obesity market by 2030, suggesting that pills could broaden uptake beyond the current 12% usage level by reaching patients less willing to use injections. ⁵
That does not mean market size by itself determines tort severity. It does mean the potential claimant pool is large, the products are highly visible, and plaintiff firms have a reason to keep investing in the space. In emerging pharmaceutical litigation, broad exposure often matters before verdict history develops. ³⁴
What We Still Don’t Know
The biggest unknown is whether the current litigation will turn into a top-tier pharmaceutical mass tort or remain a sizable but more limited wave of litigation. There are already enough cases to say the issue is real. What remains unclear is whether plaintiffs will be able to build the kind of durable scientific record that usually drives large-scale resolutions in more developed mass torts.³
That is one reason it would be premature to describe GLP-1s as the next talc or PFAS. There is not yet a reliable body of verdicts or settlement outcomes from which to estimate ultimate loss severity. For now, the litigation is easier to understand through filing volume and exposure scale than through resolution history.³
Why This Deserves Attention Now: Even without major verdicts or settlements, GLP-1 litigation already has several markers of an emerging tort risk: rapid filings, broad product use, and a growing range of alleged injuries.

Other Liability Angles Worth Watching
The GLP-1 story is not limited to traditional product liability claims against branded manufacturers. As the market expands, related issues involving promotional practices, compounded products, telehealth distribution, and broader product-risk communication may also draw scrutiny. FDA’s September 2025 warning letter to Novo Nordisk is one example of how product-risk oversight can extend beyond the tort complaints themselves.6
Emerging tort exposure does not always stay confined to the original injury allegations. It can spread into additional liability areas as demand, prescribing patterns, and product access evolve. ⁵
How Alan Gray May Be Able to Help
As GLP-1 litigation develops, insurers and other organizations may need a clearer view of claim activity, defense costs, and emerging patterns. In that setting, Alan Gray may be able to help by supporting a more disciplined approach to monitoring and managing the issue.
- Legal spend oversight: improve visibility into outside counsel activity and defense cost trends.
- Claims reporting and trend analysis: track filing activity, injury patterns, and changes in exposure over time.
- Data quality and reporting support: strengthen the underlying information used for monitoring, reserving, and management reporting.
- Early risk identification: help identify whether GLP-1 litigation remains limited or begins to develop into a broader tort exposure.
GLP-1 litigation is still developing, but it is already large enough to warrant close attention from insurers and other organizations monitoring emerging tort risk.
Citations
- United States Judicial Panel on Multidistrict Litigation. In re: Glucagon-Like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation. MDL No. 3094, Transfer Order, 2 Feb. 2024.
- United States Judicial Panel on Multidistrict Litigation. In re: Glucagon-Like Peptide-1 Receptor Agonists (GLP-1 RAs) Non-Arteritic Anterior Ischemic Optic Neuropathy Products Liability Litigation. MDL No. 3163, Transfer Order, 15 Dec. 2025.
- United States Judicial Panel on Multidistrict Litigation. Distribution of Pending MDL Dockets by Actions Pending. 1 Mar. 2024, 3 Mar. 2025, and 2 Mar. 2026.
- Lopes, Lunna, et al. “Poll: 1 in 8 Adults Say They Are Currently Taking a GLP-1 Drug for Weight Loss, Diabetes, or Another Condition, Even as Half Say the Drugs Are Difficult to Afford.” KFF, 14 Nov. 2025.
- Pradhan, Richa, and Patrick Wingrove. “Novo Nordisk Sees Pills Capturing over Third of GLP-1 Obesity Market by 2030.” Reuters, 12 Jan. 2026.
- U.S. Food and Drug Administration. “Novo Nordisk Inc. - 716495 - 09/09/2025.” Warning Letter, 9 Sept. 2025.

%20(1).png)







